Quality Improvement: Do you have the tools you need?
Fortunately, We Provide 5 Tips to Help
march 2023
By Jody Randall MSN, RN, CIC, HACP-CMS, HACP-PE
CEO and Founder
There is no shortage of quality metrics that require monitoring in healthcare. Developing and maintaining a top-notch quality program is no small task. It is also a very necessary one. It is important to remember that having a good quality plan and program in place equates to improved patient outcomes, patient satisfaction, employee satisfaction, improved efficiency, engaged leadership, and increased revenue. Here are a 5 key topics/tips to help improve your organization’s Quality Program and reap the benefits of what having a good plan and program in place can do:
- Plan: Establish or review your organization’s approved plan and program. Does your current program address safe culture, chart review, surveillance, education, risk assessments, communication, benchmarks and reporting? If not, now is a good time to correct this. Keep in mind that this should be done no less than annually. The purpose of an annual review is to determine what worked well and what could be improved.
- Oversight: It is not always easy in healthcare today to retrain or recruit experienced healthcare professionals. Practical planning is always a good strategy. Be sure that team members who oversee your quality program are educated and stay current on updates and trends. It is important for them to be knowledgeable of evidence-based practices, quality measures, gathering/review data, conducting a root cause analysis and reporting all findings to the facility’s governing body, local, state and federal agencies as appropriate.
- Change Management: It is important to consider that having a successful quality improvement program is NOT the role and responsibility of the Quality Manager or Director alone. Education and planning are essential elements of change management. A facility-wide approach is necessary in order for any quality program to be successful. Empower you team members with the tools that they need to impact change and improve the quality of care provided.
{continues after in-line diagram courtesy of CMS.gov}
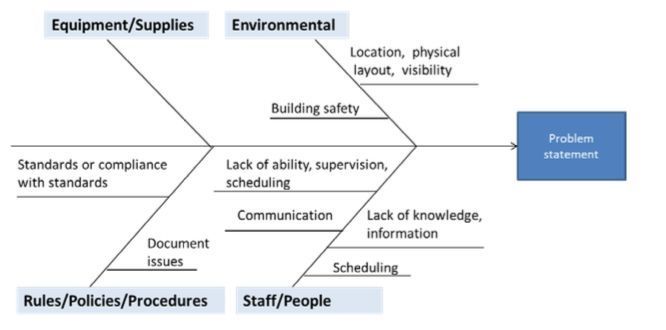
Review: While a top-notch quality plan requires a proactive approach, it is critical to conduct time sensitive reviews when an adverse event occurs. One of the easiest diagrams to use when conducting a root cause analysis (RCA) is the fishbone diagram. CMS released a revised document on how to use the fishbone tool diagram. Another tool that may be used is failure mode and effects analysis (FMEA). The RCA should be done as close to the time of the event as possible. This will help with recall of the actual events that occurred. It is important to emphasize that the review is non-punitive, and the purpose is to gather information that will be used to identify areas where process improvements may be necessary.
Reporting: Be sure to report metrics, outcomes, successes and failures in a timely manner. Delays in reporting can lead to repeat occurrences of adverse outcomes, negative impact on employee morale and a decrease in revenue. It is also important to keep in mind that your outcomes may also become public information. Specifically, if you are a CMS provider, have you looked at your facility’s data lately? Check out Medicare.gov’s Quality of Care search or The Agency for Healthcare Research and Quality’s (AHRQ) toolkit.
Worth the review, Harvard Business School published
five key steps for implementing change management. Before any healthcare organization can have a successful Quality Program, there may be some underlying changes that must occur first.
HCE is Here to Help
Healthcare Consulting Experts LLC was built based upon our understanding of the challenges that healthcare facilities are facing today. Healthcare professionals strive to deliver the best possible care to all patients. We can help your facility through the difficult times and put you back on track to a less stressful tomorrow.
Don’t take chances! Our experts can assist with regulatory compliance requirements for whether you are building a new, state-of-the-art project or renovating an existing structure. Be sure to visit
Our Website to see a full list of the services that we provide.
Contact us today at +1 (800) 813-7117 for a free initial consultation.
Please join us by clicking on any of our icons below to leave a comment or for more informati on and updates.
References:
https://www.medicare.gov/care-compare/?providerType=Hospital&redirect=true
https://www.ahrq.gov/evidencenow/tools/keydrivers/implement-qi.html